
According to the standards institute CENELEC, CE stands for Conformité Européenne. CE marking is a prerequisite for marketing products within the European Union. The product groups and technical areas concerned include, for example, the electromagnetic compatibility of equipment.
As part of our services, we offer you complete approval for the European market. We carry out the necessary tests in our own laboratories and our experienced certification team, including a notified body, paves the way for you to enter the European market with a CE mark.
To obtain a CE marking, products with radio technologies must meet the requirements defined by the European Radio Equipment Directive 2014/53/EU (RED). This directive sets the legal framework for all products with radio technologies. Key defined requirements are health and safety, electromagnetic compatibility and efficient use of radio frequencies. Manufacturers and suppliers of these products to the EU market must demonstrate compliance with RED by providing type examination based on technical documentation (TD) or declaration of conformity (DoC) and CE marking.
By definition, certification based on EN standards (EU) only provides access to EU markets. Independent certification of products with wireless technologies for other markets is therefore necessary. However, the testing effort is manageable, since a large number of the regulations for market access are based on the requirements for a CE marking. In combination with FCC certification, a large number of requirements for international market approval are therefore already covered.
Within the scope of an EU certification according to the RED, we recommend the cooperation with a notified body. As an independent organization designated by the European Commission, a notified body performs the necessary conformity assessments and supports manufacturers, especially when no harmonized standards are applicable. A notified body carries out the conformity procedure on behalf of the applicant and can verify the assessment and the corresponding type test (TCF).
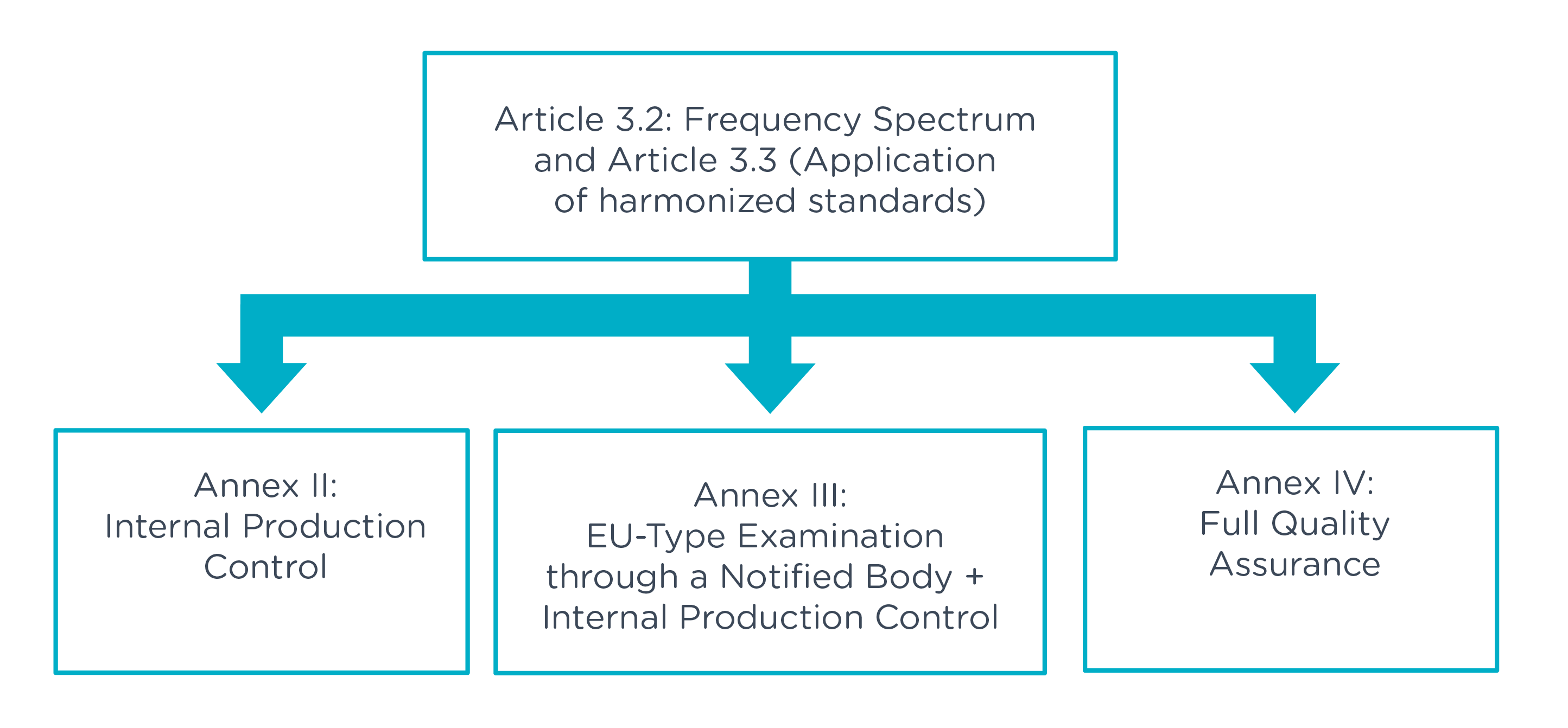
When deviating from harmonized standards or when they do not exist, a notified body can define specific tests and help define the appropriate test plans. In the case of non-harmonized (country-specific) frequency ranges, it can assist in the selection of appropriate frequency bands so that manufacturers can bring their products to market.
A notified body can handle necessary approvals for the states of the European Economic Area (EEA) according to the RED and prepares the type examination on the basis of the submitted documentation and issues a type examination certificate in the positive case.
As a notified body for various EU directives, we can support you in the CE marking process and help you gain access to the European market. Although a self-declaration procedure is allowed, some products require independent testing or certification by the notified body.

If harmonized standards are deviated from or do not exist, a notified body must be involved. If you are not familiar with the conformity assessment procedure, it is recommended to involve a notified body on a voluntary basis.
Our laboratories in Essen and Saarbrücken are accredited by the DAkkS according to DIN ISO 17025 to perform various tests for CE conformity.
Detailed information on our accreditation scope for our laboratories in Germany can be found on our accreditation overview.
For a market launch in Europe, the declaration of conformity according to the RED and the resulting application of the CE mark is essential. In addition, there are various countries that have adopted the basic principles of RED as requirements for the approval of products with radio technologies in whole or in part. The approval for the European area thus serves as an ideal starting point for potential further international market approvals.
Since 13 June 2016, Directive 2014/53/EU (RED – Radio Equipment Directive) has replaced the previous Directive (1999/5/EC). From 13 June 2017, the RED is used for CE marking of products with radio technologies.
The essential requirements are defined as follows:
Combined Equipment is the physical connection between a radio system and a device which – considered separately – falls within the scope of another directive (e.g. Machinery Directive, EMC Directive, Medical Devices Directive).This could be a washing machine with Bluetooth module or an industrial printing system with Zigbee module. Before the introduction of RED, combined equipment was often considered separately with regard to the individual components, but now the focus of RED is on the end product. In addition to the obligatory measurements regarding the radio spectrum, RED may also include other measurements that have not yet been considered for the entire device.
Generally, the measurements are the basis for issuing the declaration of conformity, as they cover the essential requirements of Article 3 of the RED. Of course these measurements can also be carried out by the manufacturer. For reasons of quality assurance (Third-Party-Opinion) we recommend, however, the involvement of an independent test house for the final conformity measurements.
Usually not, since modules are in most cases not usually tested in the (host) environment in which they are installed at the terminal device level. Tests at the terminal device level are usually unavoidable.
Usually parts of the radio tests (especially conducted test cases) can be transferred to the end device.
If the end device and USB plug-in solution are marketed separately as independent devices, they shalleach meet the requirements of the applicable directive(s). If these individual components are brought together by the end user, he is usually responsible for the resulting system. If the devices are placed on the market together (as a bundle), or the USB plug-in solution is already integrated, then the person placing the devices on the market is responsible and shallalso examine the system for its conformity.
“RED-EMC” differs from “EMC-EMC” only slightly in principle -the tests to be performed are usually the same, and the degrees of immunity may change (e.g. EMS Field Measurement : 10V/m instead of 3V/m for industrial applications). For so-called “Combined Equipment”, standards of both directives can be applied, depending on the device.
Devices that are already on the market and, in addition, no longer meet current requirements can continue to be operated after e.g. maintenance/repair-related replacement of components without having to carry out a new conformity assessment. If the replacement of components does not require special expertise and is ideally described in detail in the operating instructions (e.g. replacement of an ink cartridge in a printer), the customer can also take over the replacement.